Basic Information
Model NO.
XMHT-Diphenhydramine
Grade
Pharmaceutical Grade
Appearance
White Crystalline Powder
Certification
GMP, ISO 9001, USP, BP
Molecular Formula
C17H21NO
Product Description
Diphenhydramine is a common antihistamine medication primarily used to relieve allergy symptoms such as sneezing, runny nose, skin itching, and hives. It is also employed for the short-term management of sleep difficulties and has anti-vertigo properties. Its mechanism of action involves blocking histamine receptors in the body to reduce allergic reactions and associated discomfort.
Technical Specifications
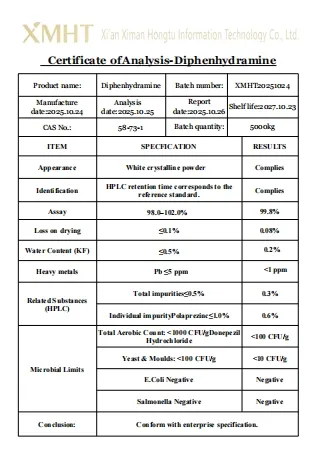
| Product Name | Diphenhydramine |
| Appearance | White Powder |
| CAS | 58-73-1 |
| MF | C17H21NO |
| MW | 291.81568 |
Application & Function
Diphenhydramine is primarily recognized for its potent antihistaminic properties, which make it highly effective in managing symptoms associated with allergic reactions. By competitively inhibiting histamine H1 receptors, it reduces or prevents typical allergic manifestations such as sneezing, rhinorrhea, pruritus, lacrimation, and urticaria.
Beyond its antihistaminic and sedative applications, diphenhydramine possesses antiemetic qualities, often employed to alleviate nausea and vomiting related to motion sickness. Its anticholinergic activity further contributes to suppressing nausea by affecting the vomiting center.
Product Display
Frequently Asked Questions (FAQ)
What is the purity of your Diphenhydramine powder?
Our Diphenhydramine powder is Pharmaceutical Grade with a purity assay of over 99% verified by HPLC testing.
What are the primary applications of this product?
It is primarily used as an antihistamine for allergy relief, a sedative for short-term sleep management, and an antiemetic for motion sickness.
Which quality certifications does the product meet?
The product complies with GMP, ISO 9001, USP, and BP standards to ensure global pharmaceutical quality.
What is the shelf life and storage condition?
The shelf life is 2 years when stored in a cool, dry place away from direct sunlight and moisture.
What is your monthly production capacity?
Our current production capacity is approximately 5000kg per month to ensure stable supply.
How is the product transported?
We offer negotiable transport packages tailored to meet safety requirements and customer needs.

























 QLU Health
QLU Health